
A stock solution is prepared by weighing out an appropriate portion of a pure solid or by measuring out an appropriate volume of a pure liquid, placing it in a suitable flask, and diluting to a known volume. Exactly how one measure’s the reagent depends on the desired concentration unit.
How do you prepare a stock solution?
- A very accurate standard alkaline solution cannot be prepared using sodium hydroxide as the primary standard. ...
- Pure anhydrous sodium carbonate, Na 2 CO 3, is used to prepare a primary standard alkaline solution.
- Solid organic acids such as oxalic acid, H 2 C 2 O 4 .2H 2 O, is used to prepare primary standard acidic solution.
How to make perfect stock?
Ingredients
- 4 pounds chicken
- 7 cups water
- 1 large onion, halved
- 3 stalks celery
- 3 carrots, cut into 2 inch pieces
- 1 bay leaf
- 1 teaspoon grated fresh ginger
- salt to taste
How to prepare stock solutions?
Solutions are often prepared by diluting a more concentrated stock solution. A known volume of the stock solution is transferred to a new container and brought to a new volume. Since the total amount of solute is the same before and after dilution, we know that. (2.5.1) C o × V o = C d × V d. where C o is the stock solution’s concentration ...
How to make an IPTG stock solution?
Stock Solution Preparation of 1 M IPTG (1 M IPTG Recipe) Dissolve 2.38 g of IPTG in 8 mL of distilled H 2 O. Bring to a final volume of 10 mL with molecular biology grade H 2 O. Filter sterilize with a 0.22 μ syringe filter. Store in 1mL aliquots at -20 °C. View this method on protocols.io and use it directly at your bench. Comparable Items:
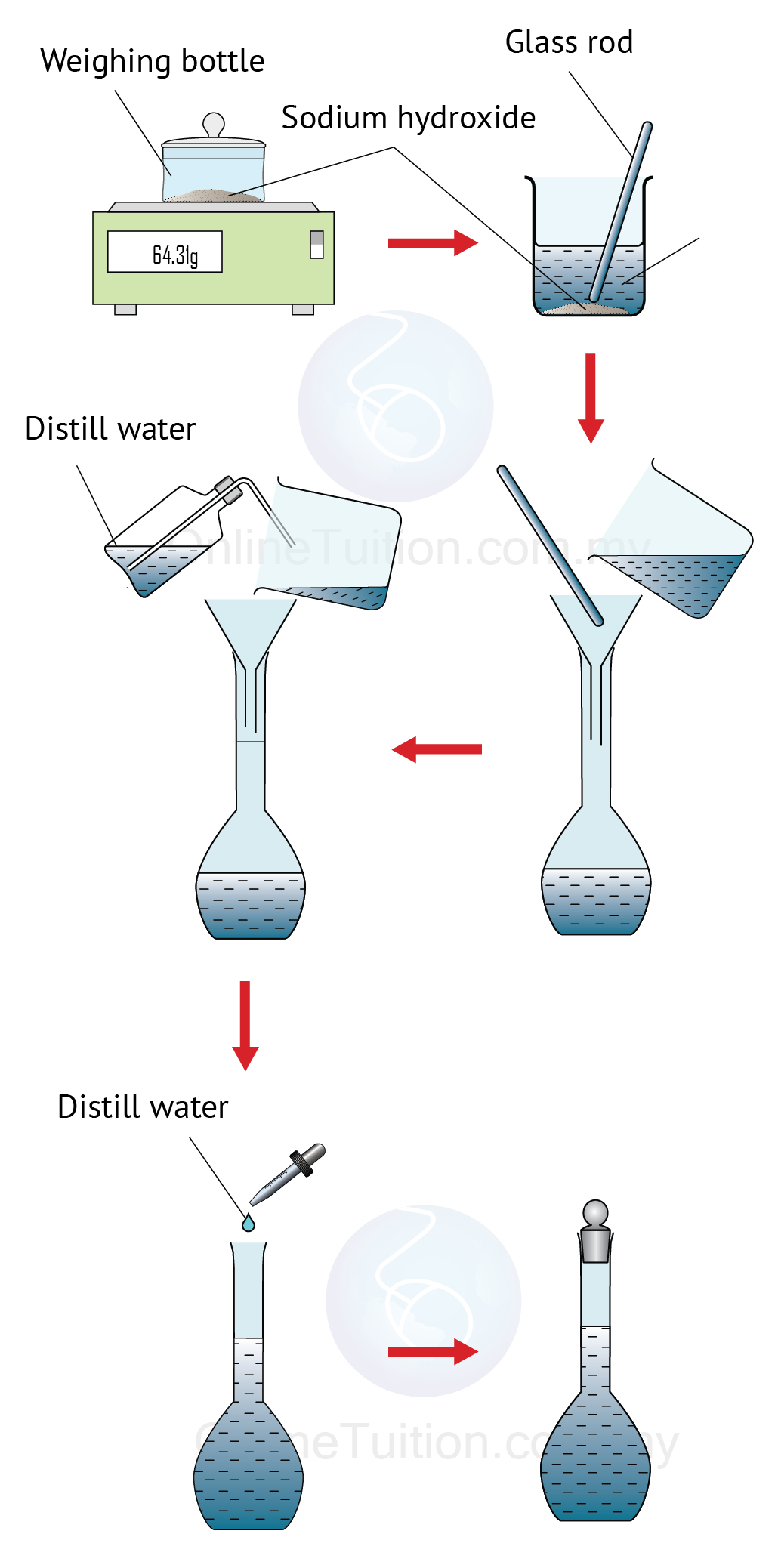
How do you prepare a solution from a solid?
From solid material Calculate the amount of solute required to prepare the desired solution. Weigh out the amount of solute calculated in step (2) and obtain a volumetric flask of appropriate volume. Add the solute to the volumetric flask. Fill the volumetric flask approximately two thirds full, stopper and mix.
How do you prepare a solution of stock?
As an example, say you need to prepare 50 milliliters of a 1.0 M solution from a 2.0 M stock solution. Your first step is to calculate the volume of stock solution that is required. To make your solution, pour 25 ml of stock solution into a 50 ml volumetric flask. Dilute it with solvent to the 50 ml line.
How do you make a molar solution from a solid?
Molar solutions are prepared by dissolving the gram molecular weight of the solute making 1 liter of solution. It means, to prepare 1 liter solution, we have to dissolve the solute equal to the molecular weight of the solute in grams. We require 98 grams of H2SO4 (100%) to prepare 1 liter of 1M H2SO4 solution.
What is a stock standard solution?
In chemistry, a stock solution is a large volume of common reagent, such as hydrochloric acid or sodium hydroxide, at a standardized concentration. This term is commonly used in analytical chemistry for procedures such as titrations, where it is important that exact concentrations of solutions are used.
How do you make stock solutions of heavy metals?
To prepare 1000 ppm stock solution , divide the molecular weight of the Salt used, by the atomic weight of concerned element, you will get a number say x then dissolve xgm of the salt into 100 ml of water and raise the volume to 1000 ml, you will get one liter of 1000 ppm solution of the concerned element.
How do you make a 10% stock solution?
How to make 10% SDS stock solutionWeigh out 10 g SDS and add to a 100 mL Duran bottle. ... Measure out 80 mL of distilled water and add to the Duran bottle.Add a magnetic flea and place on a magnetic stirring plate to mix the solution.More items...
How do you make a 0.1 M solution from a solid?
To make a 0.1M NaCl solution, you could weigh 5.844g of NaCl and dissolve it in 1 litre of water; OR 0.5844g of NaCl in 100mL of water (see animation below); OR make a 1:10 dilution of a 1M sample.
How do you make liquid solution?
Dissolve 93.52 g of NaCl in about 400 mL of distilled water, then add more water until final volume is 800 mL. If starting with a solution or liquid reagent: When diluting more concentrated solutions, decide what volume (V2) and molarity (M2) the final solution should be.
How do I make a 10 mg/ml stock solution?
To prepare a concentration of 10 µg/ml, pipette out 10 µl of the drug in a test tube using a micropipette. Then dilute it with 990 µl (making a total volume of 1 ml) of the solvent (ethanol, methanol, water etc) you are going to use in your experiment. Similarly, do for 20 µg/ml.
What is an example of a stock solution?
Examples of stock solutions are a five molar solution of NaCl or two molar solution of Tris-HCl. It is important to understand that when you are diluting a solution, you are not removing any of the solute. The total amount, or mass, of the solute does not change.
What does a 1 stock solution mean?
In preparing such solutions, keep in mind that 1% translates to 1 g/100 ml. Scale it up on the percentage (e.g. 20% means 20g/100 ml) and scale to the final volume by multiplying the values by 100 (i.e. 200g/1000ml).
How do you make a stock solution for a serial dilution?
Remember that serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. So you multiply each successive dilution by the dilution factor. You would transfer 0.2 mL from Tube 1 to 3.8 mL of diluent in Tube 2 and mix.