
The basic relationship for dilutions is C1 x V1 = C2 x V2 where C1 is initial concentration of standard solution (1000 ppm) V1 is the volume of stock solution (1ml) to be diluted to get 100 ml of 10 ppm solution C2 will be 1, 2, 5 or 10 ppm for respective dilution standards
Full Answer
How to calculate volume of stock solution for dilution?
Your first step is to calculate the volume of stock solution that is required. MdilutionVdilution = MstockVstock. (1.0 M)(50 ml) = (2.0 M)(x ml) x = [(1.0 M)(50 ml)]/2.0 M. x = 25 ml of stock solution. So to make your solution, you pour 25 ml of stock solution into a 50 ml volumetric flask.
How do you dilute a 1000 PPM stock?
Take 0.1 mL of your 1000 ppm stock and dilute it to 100 mL (use a volumetric flask for accuracy and convenience). Then, re-run the calculation using the dilution formula. To avoid waste, and depending on the volumes you need, you can proportionally cut the volume of all your stocks.
What is the dilution factor of 1 ppm?
For example, if a solution with a concentration of 1 ppm is diluted to yield a solution with a concentration of 1 ppb, the resulting dilution factor is 1000. For this particular dilution, it can also be said that the stock solution was diluted 1000- fold.
What is the formula for dilute stock solution to 5 ppb?
So, you have a stock solution of 1000000 ppb (concentration 1), and you want to know what volume (1) to dilute to make a stock solution of whatever volume you want (for example 100 mL). Thus, if you want 5 ppb, the formula is ? mL (1) x 1000000 ppb (1) = 100 mL (2) x 5 ppb (2).
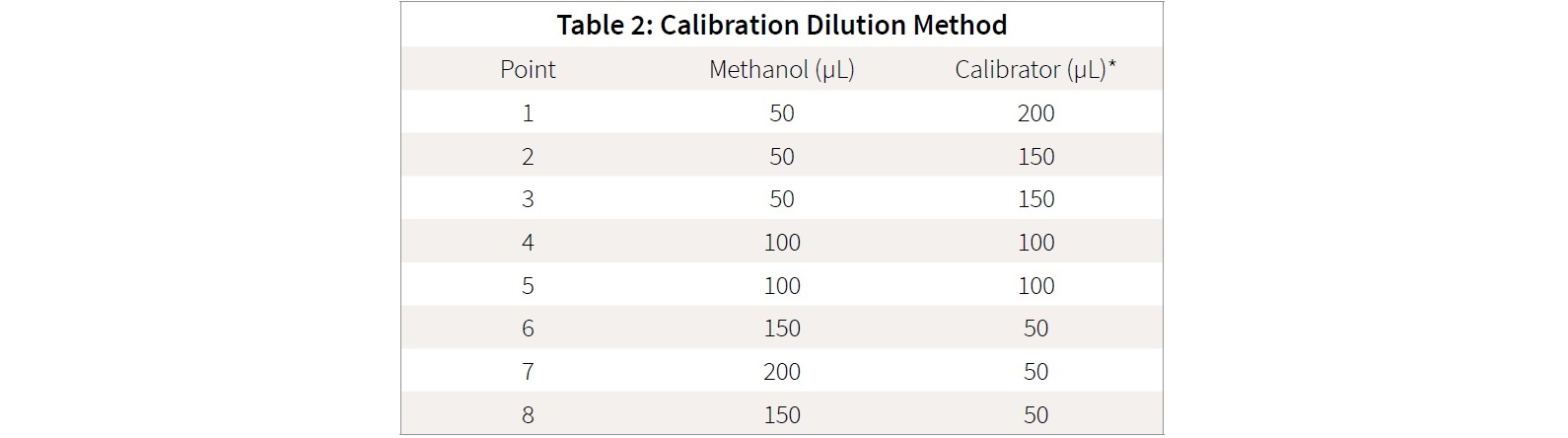
How do I make a 10ppm dilution?
One way is to take 0.5g of Liquinox dissolved in a 500 mL volumetric flask and then take 1 mL of that solution diluted to 100 mL in a 100 mL volumetric flask. This would result in 100 mL of 10 ppm Liquinox. Note that you may not need to prepare a 10 ppm Liquinox solution.
How do you dilute ppm in stock solution?
From this stock you can dilute to desired ppm of the solution. For example, if you want 100ml 1ppm solution, take 0.1mL from stock(1000 ppm) and make up to 100ml in standard measuring flask using desired solvent.
How do you dilute in ppm?
the dilute solution ? Use the dilution formula C1V1 = C2V2 where C, concentration can be in units of molarity, ppm, ppb, etc. obtain the slope for the relationship between absorbance and ppm.
How do you prepare a dilution from a stock solution?
Your first step is to calculate the volume of stock solution that is required. To make your solution, pour 25 ml of stock solution into a 50 ml volumetric flask. Dilute it with solvent to the 50 ml line.
How do you make a 10X stock solution?
To make 1 L of 10X TBS stock solution, dissolve 24 g Tris and 88 g NaCl in 900 mL of water and then adjust the pH to 7.6 and final volume to 1 L.
How do you calculate dilution?
The formula for calculating a dilution is (C1) (V1) = (C2) (V2) where...C1 is the concentration of the starting solution.V1 is the volume of the starting solution.C2 is the concentration of the final solution.V2 is the volume of the final solution.
How do you make a 5 ppm solution?
1:513:04How to prepare 5 ppm solution - YouTubeYouTubeStart of suggested clipEnd of suggested clipSolution first we take 100 mg in hundred ml of water. From this structure should between 1 ml 100 mlMoreSolution first we take 100 mg in hundred ml of water. From this structure should between 1 ml 100 ml of water.
How do you convert stock solutions to working solutions?
So you need to take 10 ml of 10 X stock solution to prepare a 100 ml working (diluted) solution. How much solvents do you need to add? So you need to mix 10 ml of 10X stock solution and 90 ml water to obtain 100 ml of working (diluted) solution.
How do I calculate ppm?
How do you calculate ppm? PPM is calculated by dividing the mass of the solute by the mass of the solution, then multiplying by 1,000,000. Both parts of the equation must be in the same format, weight or volume.
How do you dilute a stock calculator?
Dilute Solution of Known Molarity The calculator uses the formula M1V1 = M2V2 where "1" represents the concentrated conditions (i.e., stock solution molarity and volume) and "2" represents the diluted conditions (i.e., desired volume and molarity).
How do I make a 10 mg/mL stock solution?
To prepare a concentration of 10 µg/ml, pipette out 10 µl of the drug in a test tube using a micropipette. Then dilute it with 990 µl (making a total volume of 1 ml) of the solvent (ethanol, methanol, water etc) you are going to use in your experiment.
How do you make a 1/10 dilution?
For example, to make a 1:10 dilution of a 1M NaCl solution, you would mix one "part" of the 1M solution with nine "parts" of solvent (probably water), for a total of ten "parts." Therefore, 1:10 dilution means 1 part + 9 parts of water (or other diluent).
How to identify a dilution solution?
You can identify a dilution solution by the amount of solute in the total volume, expressed as a proportion. For example, a chemical may be prepared in a 1:10 dilution of alcohol, indicating that a 10 mL bottle contains one milliliter of chemical and nine milliliters of alcohol. You can calculate the necessary volume of each component to prepare a dilution solution.
How many milliliters of alcohol are in a 10 ml bottle?
For example, a chemical may be prepared in a 1:10 dilution of alcohol, indicating that a 10 mL bottle contains one milliliter of chemical and nine milliliters of alcohol. You can calculate the necessary volume of each component to prepare a dilution solution.
What is the difference between dilute solution and stock?
The only difference is that the dilute solution now contains more water than the stock from which it was prepared. In real life, what you just read is similar to you adding more water to your coffee or tea to lighten its taste. As you add more water, you are only increasing the amount of water in the solution, but not the amount ...
What does it mean to dilute a solution?
To dilute means to add a certain amount of solvent (water) to a certain amount of concentrated stock solution. If you add a certain amount of solvent to a certain amount of concentrated stock solution, you will notice that the amount of solute present in the stock solution is the same amount present in the dilute solution.
How many molecules are in a dilute solution?
Here is an illustration of a dilute solution prepared from a stock solution: From the illustration above, you can see that there are 7 molecules of solute in the concentrated solution and 7 molecules in the dilute solution. But the dilute solution has more solvent in it than the concentrated solution. Because both solutions contain an equal number ...
Which has more solvent, concentrated or dilute solution?
But the dilute solution has more solvent in it than the concentrated solution. Because both solutions contain an equal number of solute molecules, it follows that: the moles of chemicals present in the concentrated solution is equal to the moles of chemicals present in the dilute solution. If we translate the previous statement into ...
What is stock solution?
posted on February 10, 2019. A stock or standard solution is a solution in which you accurately know its concentration. You can make stock solutions in the chemistry laboratory or buy from chemical manufacturers. Once you have a stock solution, you can prepare solutions of lower concentration by diluting the concentrated stock solution.
Why do we add sulfuric acid to water?
We did this in order to prevent the solution from exploding. As you may know, sulfur ic acid is much denser than water, so as you add it to water, its molecules are able to travel within and mix well with the water molecules. However, because water is less dense than sulfuric acid, if you add it to sulfuric acid, ...
How to do a dilution in chemistry?
Performing a dilution in chemistry usually means taking a small amount of a solution whose concentration you know, then adding a neutral liquid (like water) to make a new solution with a larger volume but a lower concentration.
What is dilution in science?
Dilution is the process of making a concentrated solution less concentrated. There are a variety of reasons why one might want to perform a dilution. For example, biochemists dilute solutions from their concentrated form to create new solutions for use in their experiments. As a more casual type of dilution, bartenders often dilute hard liquor ...
How much water to dilute orange juice?
For example, if you want to dilute 1 cup of concentrated orange juice to ¼ its initial concentration, add 3 cups of water for a total of 4 cups liquid . To learn how to accurately dilute concentrates with dilution equations, keep reading!
Why are dilutions in different units?
Because dilutions involve changes in concentration (which can sometimes be quite large), it's not uncommon for two variables in your equation to be in different units. Though this is easily overlooked, mismatching units in your equation can cause your answer to be orders of magnitude off.
What happens when you add powder to a liquid?
In other words, when adding small amounts of powder to a liquid, just add the powder to the final volume of liquid you want and mix.
Do you know the concentration of a solution?
Usually, in most real-world situations, you will know the concentration of your starting solution and both the concentration and volume you want in your second solution, but not the volume of the first solution you need to use to get there.
Is orange juice diluted?
For instance, the simple act of making orange juice from concentrate is a dilution. In many cases, products that need to be diluted include information about the dilution that needs to be made somewhere on the packaging. They may even have precise directions to follow.
Popular Answers (1)
Use the standard dilution formula in general chemistry, i.e., Volume (1) x Concentration (1) = Volume (2) x Concentration (2). Make sure all your units are the same. Thus, you can either change 1000 ppm to ppb (1000 ppm x 1000 ppb/1ppm) = 1000000 ppb or you can change the ppb needed to ppm.
All Answers (10)
Use the standard dilution formula in general chemistry, i.e., Volume (1) x Concentration (1) = Volume (2) x Concentration (2). Make sure all your units are the same. Thus, you can either change 1000 ppm to ppb (1000 ppm x 1000 ppb/1ppm) = 1000000 ppb or you can change the ppb needed to ppm.
Similar questions and discussions
Can anyone suggest a simple calculation procedure to prepare any solution in terms of ppm ?
