
Multiply the final desired volume by the dilution factor to determine the needed volume of the stock solution. In our example, 30 mL x 1 ÷ 20 = 1.5 mL of stock solution. Subtract this figure from the final desired volume to calculate the volume of diluent required--for example, 30 mL - 1.5 mL = 28.5 mL.
How to calculate volume of stock solution for dilution?
Your first step is to calculate the volume of stock solution that is required. MdilutionVdilution = MstockVstock. (1.0 M)(50 ml) = (2.0 M)(x ml) x = [(1.0 M)(50 ml)]/2.0 M. x = 25 ml of stock solution. So to make your solution, you pour 25 ml of stock solution into a 50 ml volumetric flask.
How do I make a stock solution?
Your first step is to calculate the volume of stock solution that is required. To make your solution, pour 25 ml of stock solution into a 50 ml volumetric flask. Dilute it with solvent to the 50 ml line.
How do you calculate dilution factor?
For example, a 1:20 dilution converts to a 1/20 dilution factor. Multiply the final desired volume by the dilution factor to determine the needed volume of the stock solution. In our example, 30 mL x 1 ÷ 20 = 1.5 mL of stock solution. Subtract this figure from the final desired volume to calculate the volume of diluent required--for example, ...
How to calculate volume of concentrated stock solution (Vol con)?
Since we need the volume of concentrated stock solution (Vol con ), we must divide both the left and right side of the equal sign in the above equation (1) by 5 M. If we do, we will get: From the calculation, you need to pipette 4 mL of the 5 M sulfuric acid solution to prepare 10 mL of 2 M sulfuric acid solution.
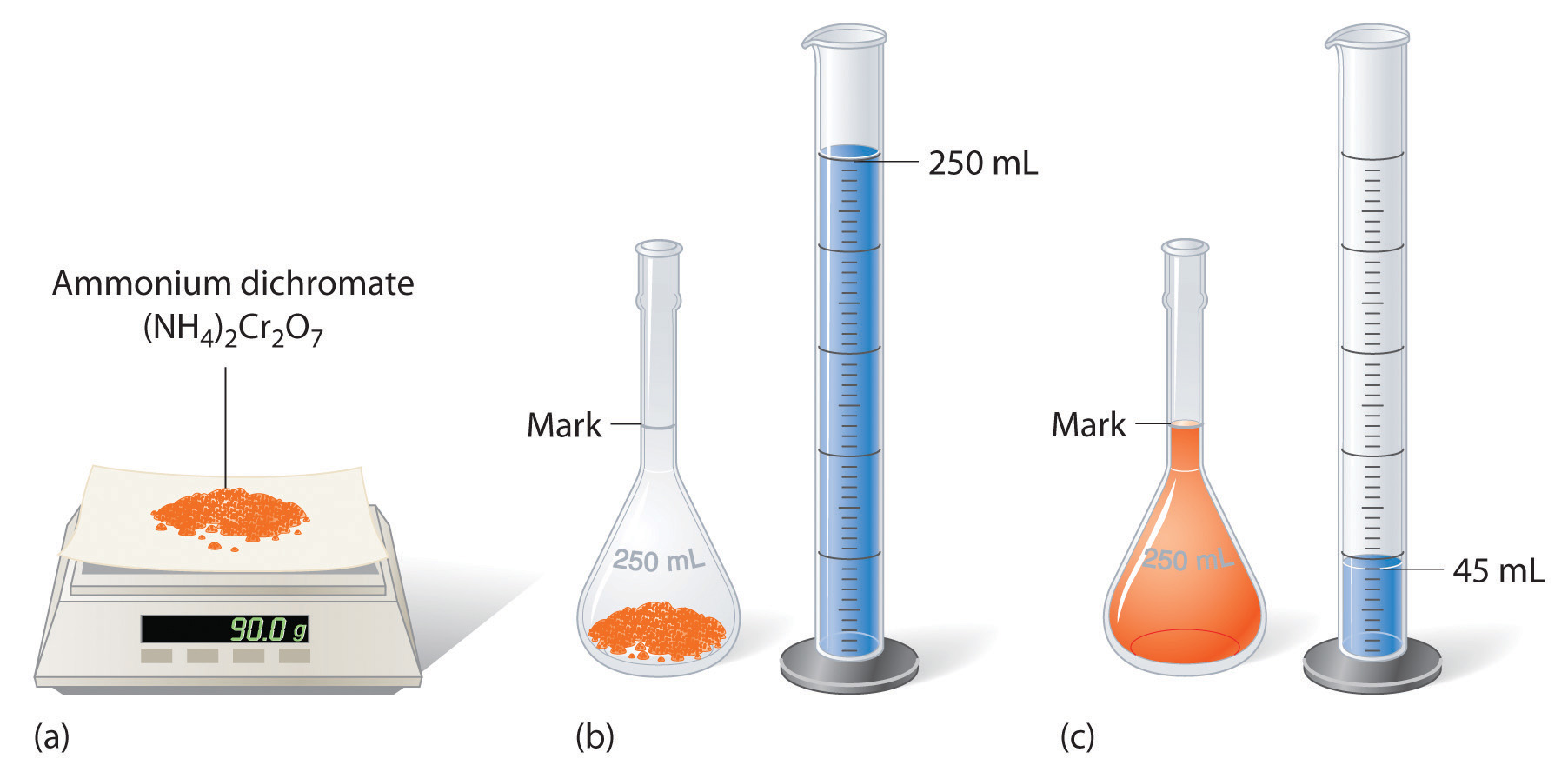
How do you calculate volume of water needed?
Multiply length (L) by width (W) to get area (A). Multiply area by height (H) to get volume (V). Multiply volume by 7.48 gallons per cubic foot to get capacity (C).
How do you find the volume of a stock solution?
To determine the volume of stock solution needed, divide the number of moles of glucose by the molarity of the stock solution.
How do you calculate the amount of water in a solution?
1:074:06Calculating the amount of water needed to be added to a stock ...YouTubeStart of suggested clipEnd of suggested clipSo the easiest starting point is to use our trusted c1v1 equals c2v2 equation and here this would beMoreSo the easiest starting point is to use our trusted c1v1 equals c2v2 equation and here this would be everything from our stock.
How do you calculate volume needed?
Whereas the basic formula for the area of a rectangular shape is length × width, the basic formula for volume is length × width × height. How you refer to the different dimensions does not change the calculation: you may, for example, use 'depth' instead of 'height'.
How much stock solution and diluent are needed?
Multiply the final desired volume by the dilution factor to determine the needed volume of the stock solution. In our example, 30 mL x 1 ÷ 20 = 1.5 mL of stock solution. Subtract this figure from the final desired volume to calculate the volume of diluent required--for example, 30 mL - 1.5 mL = 28.5 mL.
How do you calculate the volume needed to dilute a solution?
The formula for calculating a dilution is (C1) (V1) = (C2) (V2) where...C1 is the concentration of the starting solution.V1 is the volume of the starting solution.C2 is the concentration of the final solution.V2 is the volume of the final solution.
How do you calculate the volume of water in a bucket?
2:045:57Calculate Amount of Water in a Bucket - YouTubeYouTubeStart of suggested clipEnd of suggested clipArea of the bucket multiplied by height. So we multiplied an area with the height. This will giveMoreArea of the bucket multiplied by height. So we multiplied an area with the height. This will give you the volume.
How do you calculate the volume of water in a tube?
The formula for the volume of cylinder is: cylinder volume = π * radius² * height . For a pipe use its length instead of height: pipe volume = π * radius² * length , where radius = inner diameter/2 . The volume of a pipe is equal to the volume of a liquid inside (if a pipe is fully filled with it).
How do you calculate water volume in litres?
Dividing the volume (in cubic centimeters) of the shape by 1,000 will give you the volume in liters (L). . So, a fish tank that is 40.64 cm long, 25.4 cm wide, and 20.32 tall has a volume of 20.975 L.
How do you dilute a stock solution with water?
1:243:35How to Dilute a Solution - YouTubeYouTubeStart of suggested clipEnd of suggested clipPlace the pipette in the liquid to be collected. Squeeze the pipette bulb and allow the liquid toMorePlace the pipette in the liquid to be collected. Squeeze the pipette bulb and allow the liquid to rise up the pipette. You should allow the liquid to pass the line marked on the pipette.
Why do you start with a concentrated solution and then dilute it to make a dilution?
The primary reason you start with a concentrated solution and then dilute it to make a dilution is that it's very difficult—and sometimes impossible—to accurately measure solute to prepare a dilute solution, so there would be a large degree of error in the concentration value.
What is a dilute solution?
A dilution is a solution made by adding more solvent to a more concentrated solution (stock solution), which reduces the concentration of the solute. An example of a dilute solution is tap water, which is mostly water (solvent), with a small amount of dissolved minerals and gasses (solutes).
Can you dilute a solution with too much solvent?
It's a common mistake to add too much solvent when making the dilution. Make sure you pour the concentrated solution into the flask and then dilute it to the volume mark. Do not, for example, mix 250 ml of concentrated solution with 1 liter of solvent to make a 1-liter solution.
What is the difference between dilute solution and stock?
The only difference is that the dilute solution now contains more water than the stock from which it was prepared. In real life, what you just read is similar to you adding more water to your coffee or tea to lighten its taste. As you add more water, you are only increasing the amount of water in the solution, but not the amount ...
What is stock solution?
posted on February 10, 2019. A stock or standard solution is a solution in which you accurately know its concentration. You can make stock solutions in the chemistry laboratory or buy from chemical manufacturers. Once you have a stock solution, you can prepare solutions of lower concentration by diluting the concentrated stock solution.
How many molecules are in a dilute solution?
Here is an illustration of a dilute solution prepared from a stock solution: From the illustration above, you can see that there are 7 molecules of solute in the concentrated solution and 7 molecules in the dilute solution. But the dilute solution has more solvent in it than the concentrated solution. Because both solutions contain an equal number ...
What does it mean to dilute a solution?
To dilute means to add a certain amount of solvent (water) to a certain amount of concentrated stock solution. If you add a certain amount of solvent to a certain amount of concentrated stock solution, you will notice that the amount of solute present in the stock solution is the same amount present in the dilute solution.
Why does water react with sulfuric acid?
However, because water is less dense than sulfuric acid, if you add it to sulfuric acid, its molecules will create a barrier in which you have water on top and sulfuric acid below. This unwanted barrier can cause the two chemicals to react explosively ( exothermic reaction ), generating enough energy to break the flask.
Why do we add sulfuric acid to water?
We did this in order to prevent the solution from exploding. As you may know, sulfur ic acid is much denser than water, so as you add it to water, its molecules are able to travel within and mix well with the water molecules. However, because water is less dense than sulfuric acid, if you add it to sulfuric acid, ...
Which has more solvent, concentrated or dilute solution?
But the dilute solution has more solvent in it than the concentrated solution. Because both solutions contain an equal number of solute molecules, it follows that: the moles of chemicals present in the concentrated solution is equal to the moles of chemicals present in the dilute solution. If we translate the previous statement into ...
How to identify a dilution solution?
You can identify a dilution solution by the amount of solute in the total volume, expressed as a proportion. For example, a chemical may be prepared in a 1:10 dilution of alcohol, indicating that a 10 mL bottle contains one milliliter of chemical and nine milliliters of alcohol. You can calculate the necessary volume of each component to prepare a dilution solution.
How many milliliters of alcohol are in a 10 ml bottle?
For example, a chemical may be prepared in a 1:10 dilution of alcohol, indicating that a 10 mL bottle contains one milliliter of chemical and nine milliliters of alcohol. You can calculate the necessary volume of each component to prepare a dilution solution.
